In 1907, Rutherford returned to England, transferring to a professorship at the University of Manchester. Achieving fame for his contributions to the understanding of radioelements, Rutherford became an active public speaker, published numerous magazine articles and wrote the most highly regarded textbook of the time on radioactivity. 
Rutherford and Yale Professor Bertram Borden Boltwood went on to categorize radioactive elements into what they called a “decay series.” Rutherford was also credited with discovering the radioactive gas radon while at McGill. 
At McGill in 1903, Rutherford and has colleague Frederick Soddy introduced their disintegration theory of radioactivity, which claimed radioactive energy was emitted from within an atom and that when alpha and beta particles were emitted at the same time, they caused a chemical change across elements. Rutherford left Cambridge in 1902 and took up a professorship at McGill University in Montreal. The beta particle was, in fact, the same as an electron or positron. He labeled the two radiation types “alpha” and “beta.” As it turns out, the alpha particle was identical to the nucleus of a helium atom. While Thomson went on to examine what would later be called an electron, Rutherford took a closer look at ion-producing radiations.įocusing on uranium, Rutherford discovered that placing it near foil resulted in one type of radiation being easily soaked up or blocked, while a different type had no trouble penetrating the same foil. Together, Rutherford and Thomson studied the effects of X-rays on the conductivity of gases, resulting in a paper about dividing atoms and molecules into ions. German physicist Wilhelm Conrad Röntgen had discovered X-rays just months before Rutherford arrived at Cavendish, and X-rays were a hot topic among research scientists. Thomson to collaborate on a study of X-rays. In 1895, as the first research student at the University of Cambridge’s Cavendish Laboratory in London, Rutherford identified a simpler and more commercially viable means of detecting radio waves than had been previously established by German physicist Heinrich Hertz.Īlso while at Cavendish Laboratory, Rutherford was invited by Professor J.J. 
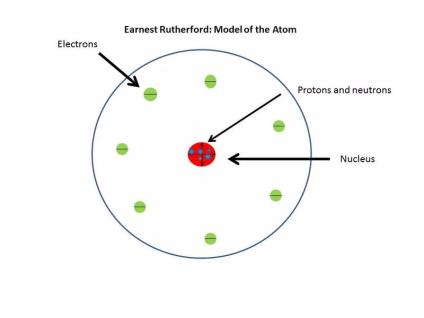
The couple married in 1900 and later welcomed a daughter, whom they named Eileen. During that same year, Rutherford met and fell in love with his landlady’s daughter, Mary Newton. His research earned him a Bachelor of Science degree in just one year’s time. In 1894, still at Canterbury, Rutherford conducted independent research on the ability of high-frequency electrical discharge to magnetize iron. Rutherford obtained both his Bachelor of Arts and his Master of Arts degrees there, and managed to achieve first-class honors in math and science. At Canterbury College, Rutherford’s professors fueled his enthusiasm for seeking concrete proof through scientific experimentation. In 1890 Rutherford landed another scholarship-this time to Canterbury College in Christchurch, New Zealand. In 1887 he was awarded a scholarship to attend Nelson Collegiate School, a private secondary school where he would board and play rugby until 1889. Despite the outcome, Rutherford’s interest in academics remained unfaltering. The young Rutherford constructed a miniature cannon, which, to his family’s surprise, promptly and unexpectedly exploded. It was a pivotal moment for Rutherford, given that the book inspired his very first scientific experiment. “We haven’t the money, so we’ve got to think,” was Rutherford’s motto at the time.Īt the age of 10, Rutherford was handed his first science book, at Foxhill School. Since money was tight, Rutherford found inventive ways of overcoming his family’s financial challenges, including birds-nesting to earn funds for his kite-flying supplies. Weekends were spent swimming in the creek with his brothers. She believed that knowledge was power, and placed a strong emphasis on her children’s education.Īs a child, Ernest, whose family called him “Ern,” spent most of his time after school milking cows and helping with other chores on the family farm. Ernest’s mother, Martha, worked as a schoolteacher. His father, James, had little education and struggled to support the large family on a flax millers' income. He was the fourth of 12 children and the second son. Early Life and EducationĮrnest Rutherford was born in rural Spring Grove, on the South Island of New Zealand on August 30, 1871. Dubbed the “Father of the Nuclear Age,” Rutherford died in Cambridge, England, on October 19, 1937, of a strangulated hernia. A pioneer of nuclear physics and the first to split the atom, Ernest Rutherford was awarded the 1908 Nobel Prize in Chemistry for his theory of atomic structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
